Riegel v. Medtronic, Inc.
United States Supreme Court
552 U.S. 312 (2008)

- Written by Sean Carroll, JD
Facts
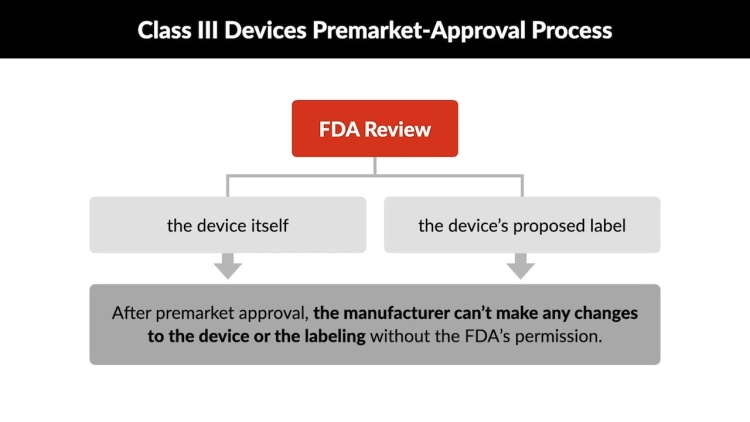
Charles Riegel and his wife (plaintiffs) brought a products-liability suit against Medtronic, Inc. (defendant) after a balloon catheter manufactured by Medtronic ruptured in Riegel’s coronary artery during heart surgery. Medtronic had received premarket approval for the catheter from the Food and Drug Administration (FDA). The premarket approval process is extensive and includes detailed review of a device's safety and effectiveness. Once the FDA has given premarket approval for a device, the manufacturer may not make any changes to the device that would affect its safety or effectiveness without the FDA's permission. In their suit, the Riegels asserted a variety of New York State common-law claims based on allegations that the Medtronic catheter was designed, labeled, and manufactured in a way that violated New York common law. The trial court awarded summary judgment to Medtronic, finding that the state claims were preempted by the federal Food, Drug, and Cosmetic Act (FDCA). The Medical Device Amendments (MDA) to the FDCA provided, among other things, that a state may not establish or use any premarket safety approval requirement for medical devices that were “different from, or in addition to, any requirement applicable . . . to the device” under federal law relating to the device's safety or effectiveness. The United States Court of Appeals for the Second Circuit affirmed the grant of summary judgment. The United States Supreme Court granted certiorari.
Rule of Law
Issue
Holding and Reasoning (Scalia, J.)
Dissent (Ginsburg, J.)
What to do next…
Here's why 921,000 law students have relied on our case briefs:
- Written by law professors and practitioners, not other law students. 47,300 briefs, keyed to 1,000 casebooks. Top-notch customer support.
- The right amount of information, includes the facts, issues, rule of law, holding and reasoning, and any concurrences and dissents.
- Access in your classes, works on your mobile and tablet. Massive library of related video lessons and high quality multiple-choice questions.
- Easy to use, uniform format for every case brief. Written in plain English, not in legalese. Our briefs summarize and simplify; they don’t just repeat the court’s language.